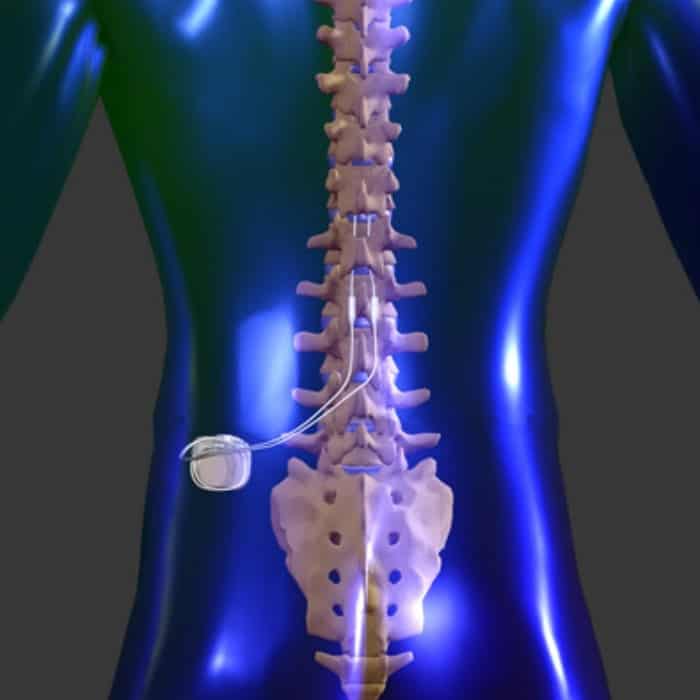
Food and Drug Administration (FDA) has approved its new Proclaim Plus spinal cord stimulation. Dank der neuesten Fortschritte von Abbott haben Ärzte nunmehr Zugang zu zwei weiteren. 23, 2022 /PRNewswire/ - Abbott (NYSE: ABT) today announced that the U.S. In der Vergangenheit hatten Ärzte ausschließlich Zugang zu einer Form der Neurostimulation, der traditionellen tonischen Rückenmarkstimulation (Spinal Cord Stimulation, SCS). Dabei kommt ein sogenannter Neurostimulator bzw. Refer to the IFU for additional information. Traditionelle neurostimulation und neurostimulation von abbott im vergleich. SCS) ist ein minimalinvasives operatives Verfahren zur Behandlung chronischer Schmerzen, welches in der Neurochirurgie der Beta Klinik Bonn angewandt wird. Safety comparisons and specific dose-response curves for each dosage have not been clinically established. NOTE: In neurostimulation therapy, ‘dose’ refers to the delivery of a quantity of energy to tissue. *Up to 10 years of battery longevity at the lowest dose setting: 0.6mA, 500 Ohms, duty cycle 30s on/360s off. The Abbott Proclaim XR SCS System benefits include 26, 2023 /PRNewswire/ - Abbott (NYSE: ABT) announced today that the U.S.The Abbott Proclaim XR can seamlessly fit into a patient’s lifestyle by allowing them to discreetly manage their pain by using familiar Apple mobile digital devices and Bluetooth wireless technology. Background context: Psychological characteristics such as catastrophizing and depression have been shown to negatively impact outcome prognosis after spinal interventions.The Abbott Proclaim XR SCS System is the latest patient-centric innovation exclusively from Abbott.At the core of this patient-centric advancement is a battery that can last up to 10 years at low-dose settings* without the hassle of recharging.Spinal cord stimulation can improve overall quality of life and sleep, and reduce the need for pain medicines. Spinal cord stimulators require two procedures to test and implant the device: the trial and the implantation. Provides pain relief without the hassle of recharging Spinal cord stimulation is used most often after nonsurgical pain treatment options have failed to provide sufficient relief.Can be dosed without sacrificing efficacy Food and Drug Administration (FDA) approved Abbotts Eterna spinal cord stimulation (SCS) system for the treatment of chronic pain This neuromodulation device provides an optimized.The Abbott Proclaim XR SCS system harnesses the power of low-energy BurstDR stimulation coupled with BoldXR™ dosing protocol that Leider lässt der Beitrag eine wichtige evidenzbasierte Therapieoption aus: die elektrische Rückenmarkstimulation (spinal cord stimulation, SCS).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
